Sodium Thioglycolate
Price 3000 INR/ Kilograms
MOQ : 1 Kilograms
Sodium Thioglycolate Specification
- Molecular Weight
- 114.094 Grams (g)
- Physical Form
- Liquid
- Molecular Formula
- C2H3NaO2S
- Shelf Life
- 1-3 Years
- Chemical Name
- Sodium Thioglycolate
- Type
- Chemical
- Purity(%)
- 98 %
Sodium Thioglycolate Trade Information
- Minimum Order Quantity
- 1 Kilograms
- Payment Terms
- Cash Against Delivery (CAD), Cash on Delivery (COD), Paypal, Letter of Credit (L/C), Western Union, Letter of Credit at Sight (Sight L/C), Telegraphic Transfer (T/T), Delivery Point (DP), Days after Acceptance (DA), Cash in Advance (CID), Cheque, Cash Advance (CA)
- Supply Ability
- 10 Kilograms Per Day
- Delivery Time
- 2-3 Days
- Sample Available
- Yes
- Sample Policy
- Free samples are available
- Main Export Market(s)
- Middle East, Africa, Australia, Central America, North America, South America, Eastern Europe, Western Europe, Asia
- Main Domestic Market
- All India
About Sodium Thioglycolate
Sodium Thioglycolate is a white solid chemical compound which acts as a reducing agent and consist of a thioglycolate(1-). It is hygroscopic in nature and is harmful and irritant in nature and can be stored at a temperature of -20 degree Celsius. It is mostly used as reducing agent for anaerobic and microaerophilic bacterial growth. Sodium Thioglycolate can also be used as a dipilatory, irritant and chelating agent. It has a chemical formula of C2H3NaO2S with a molecular weight of 114.1grams per mol. This compound is corrosive and acutely toxic in nature which may cause skin burns and severe eye damage.Sodium Thioglycolate Key Features: Usage in cold hair waving Best functions as a depilatory and reducing agent Accepts 3 and donates 1 hydrogen bond Colorless with the blunt irritating smell Product Details: Product Name : Sodium Thioglycolate Chemical Names : SODIUM THIOGLYCOLATE; 367-51-1; Sodium mercaptoacetate; Mercaptoacetic acid sodium salt; Mollescal SF; Sodium 2-sulfanylacetate Molecular Formula : C2H3NaO2S Molecular Weight : 114.094 g/mol IUPAC Name : sodium;2-sulfanylacetate PubChem CID : 23690444 CAS No : 367-51-1 EC Number : 206-696-4 Monoisotopic Mass : 113.975 g/mol Exact Mass : 113.975 g/mol Physical Description : It is a white powder with a slight odor. Used in cold-waving of hair and as a dipilatory. Melting Point : greater than 572 F Solubility : Very soluble Air and Water Reactions : Hygroscopic. Discolors on exposure to air. Water soluble.FAQs of Sodium Thioglycolate:
Q: What is the physical form of Sodium Thioglycolate?
A: The physical form of Sodium Thioglycolate is liquid.Q: What is the purity percentage of Sodium Thioglycolate?
A: The purity of Sodium Thioglycolate is 98%.Q: What is the molecular weight of Sodium Thioglycolate?
A: The molecular weight of Sodium Thioglycolate is 114.094 grams (g).Q: What is the molecular formula of Sodium Thioglycolate?
A: The molecular formula of Sodium Thioglycolate is C2H3NaO2S.Q: What is the shelf life of Sodium Thioglycolate?
A: The shelf life of Sodium Thioglycolate is 1-3 years.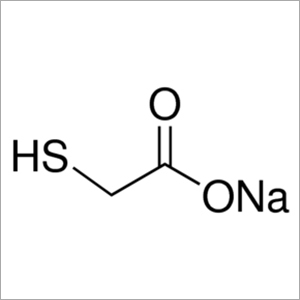
Tell us about your requirement

Price:
Quantity
Select Unit
- 50
- 100
- 200
- 250
- 500
- 1000+
Additional detail
Mobile number
Email
More Products in Laboratory Reagents Category
Carbon Tetrachloride
Type : Organic Compound
Storage : Store in a cool dry wellventilated area away from heat and ignition sources, Other
Physical Form : Liquid
Purity(%) : 99%
Strontium Bromide LR
Minimum Order Quantity : 1 Kilograms
Type : Chemical
Storage : Dry Place
Physical Form : Powder
Purity(%) : 98 %
Chloramine T LR/AR
Minimum Order Quantity : 1 Kilograms
Type : Chemical
Storage : Dry Place
Physical Form : Powder
Purity(%) : 97 %
Peptone Powder
Price 600.0 INR / Kilograms
Minimum Order Quantity : 25 Kilograms
Type : Peptone Powder
Storage : Room Temperature
Physical Form : Powder
Purity(%) : High
 |
SUVIDHINATH LABORATORIES
All Rights Reserved.(Terms of Use) Developed and Managed by Infocom Network Private Limited. |




 Send Inquiry
Send Inquiry